China Innovative Drug NHWD-870 Receives Breakthrough Therapy Designation, Bringing New Hope of Survival to NUT Carcinoma Patients
SHAOXING, China, May 9, 2026 /PRNewswire/ — Recently, the novel oral BET inhibitor NHWD-870 HCI, independently developed by Zhejiang Wenda Pharmaceutical Technology Co., Ltd., officially received Breakthrough Therapy Designation from the China National Medical Products Administration (NMPA) for the proposed indication of advanced thoracic midline (NUT) carcinoma in patients who have failed prior chemotherapy. This milestone progress brings new targeted therapy hope to NUT carcinoma patients, a disease with extremely high malignancy and a very poor prognosis.
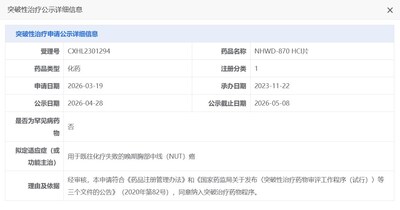
NUT carcinoma is a rare, poorly differentiated carcinoma characterized by NUTM1 gene rearrangement. The disease progresses rapidly, with a median survival of only approximately 6.5 months. It commonly occurs in the lungs, head, and neck regions, and the median age of onset is only 23.6 years. Due to the lack of specificity in clinical manifestations and insufficient awareness among the public, the rates of misdiagnosis and missed diagnosis are extremely high, and most patients are already at an advanced stage at the time of diagnosis. Currently, no effective targeted drug for NUT carcinoma has been approved globally, and the treatment landscape is extremely dire.
The granting of Breakthrough Therapy Designation to NHWD-870 HCI is primarily based on the significant efficacy and survival benefits demonstrated in its Phase II clinical study. As of December 27, 2025, a total of 40 evaluable subjects with advanced NUT carcinoma were enrolled in the study. The data showed that the drug exhibited positive anti-tumor activity across different lesion sites: the objective response rate (ORR) in 20 patients with advanced thoracic NUT carcinoma reached as high as 45.00%; the median overall survival (mOS) for advanced thoracic NUT carcinoma patients and for all subjects both reached 9.33 months, representing a significant extension compared with standard chemotherapy regimens. Currently, some patients are still receiving ongoing treatment and close follow-up. In addition, preliminary safety data showed that the drug was generally well tolerated and held high potential for clinical application.
This Breakthrough Therapy Designation is of great significance for NUT carcinoma patients and represents a leapfrog advance in the field. For patients, it means that the regulatory review process for NHWD-870 HCI will be accelerated and the drug will reach the market sooner, enabling patients with advanced NUT carcinoma to receive targeted therapy earlier and escape the dilemma of having no effective drug, thereby significantly prolonging survival and improving quality of life. On the clinical level, the development and designation of this drug fill the gap in targeted treatment for NUT carcinoma and provide clinicians with a completely new treatment option. This not only drives the transformation of NUT carcinoma treatment from “symptomatic treatment” to “precision targeted therapy” but also provides valuable clinical experience for the future development of similar drugs.
Mr. Nenghui Wang, Founder of Wenda Pharma, stated: “We are delighted that our product has received Breakthrough Therapy Designation. This progress highlights China’s growing capabilities in innovative drug development for rare cancers. NHWD-870 not only brings hope to NUT carcinoma patients but also offers new avenues for exploring treatments for other rare malignancies.”
Professor YIN Mingzhu, Director of the Midline (NUT) Carcinoma Specialty at the Affiliated Three Gorges Hospital of Chongqing University, noted, “NUT carcinoma once plunged countless patients and doctors into despair. The Breakthrough Therapy Designation for NHWD-870 is an important achievement integrating medical research and clinical practice. In the future, with the accelerated market entry of this drug, the treatment landscape of NUT carcinoma is expected to be completely transformed, injecting new momentum into the cause of rare disease diagnosis and treatment in our country.”
About Wenda Pharma
Founded in 2013 and headquartered in Shaoxing City, China. Wenda Pharma is a company dedicated to pioneering, innovative, and high-quality drug discovery. The company focuses its research in the areas of oncology, immunology, and neurodegenerative diseases, with multiple Chinese and global patent applications and grants in these fields. Several of the company’s drug candidates are currently in Phase I, II, and III clinical trials.
The NHWD-870 program, targeting the rare NUT carcinoma, has successfully completed Phase II clinical trials and holds the potential to become a “First-in-Class” oral small-molecule BET inhibitor, offering patients the hope of extended survival. Additionally, the Company’s WD-890 TYK2 (JH2) inhibitor—indicated for moderate-to-severe plaque psoriasis—has entered Phase III clinical trials, while Phase II trials for the systemic lupus erythematosus (SLE) indication are also set to commence shortly. WD-910, a therapeutic candidate targeting neurodegenerative diseases, is poised to enter Phase II clinical trials in China, which just completed phase I clinical trial in Australia, its 100% blood-brain barrier penetration capability positions it as a potential “Best-in-Class” drug candidate. Wenda Pharma boasts a rich and innovative pipeline targeting numerous refractory and incurable diseases, striving to provide patients with more effective, safe, and cost-effective innovative medicines.
www.wendapharma.com
For BD and Investors: [email protected]
Photo – https://mma.prnewswire.com/media/2975731/20260508174803_57_324.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/china-innovative-drug-nhwd-870-receives-breakthrough-therapy-designation-bringing-new-hope-of-survival-to-nut-carcinoma-patients-302766935.html
View original content:https://www.prnewswire.co.uk/news-releases/china-innovative-drug-nhwd-870-receives-breakthrough-therapy-designation-bringing-new-hope-of-survival-to-nut-carcinoma-patients-302766935.html
 Disclaimer: The above press release comes to you under an arrangement with PR Newswire. UK Newshour takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with PR Newswire. UK Newshour takes no editorial responsibility for the same.

