Preliminary Results for the Year Ended 31 December 2025

March 27, 2026
Biodexa Pharmaceuticals PLC
(“Biodexa” or the “Company” or, together with its subsidiaries, the “Group”)
Preliminary Results for the Year Ended 31 December 2025
Biodexa Pharmaceuticals PLC (Nasdaq: BDRX), a clinical-stage biopharmaceutical company developing innovative products focused on the treatment or prevention of gastrointestinal cancers, announces its audited preliminary results for the year ended 31 December 2025.
For more information, please contact:
Biodexa Pharmaceuticals PLC
Stephen Stamp, CEO
Fiona Sharp, CFO
Tel: +44 (0)29 2048 0180
www.biodexapharma.com
About Biodexa
The Company’s lead development programs include eRapa, under development for Familial Adenomatous Polyposis and Non-Muscle Invasive Bladder Cancer, MTX240 under development for Gastrointestinal Stromal Tumors (GIST) and tolimidone, under development for the treatment of type 1 diabetes.
eRapa is a proprietary oral capsule formulation of rapamycin, also known as sirolimus. Rapamycin is an mTOR (mammalian Target Of Rapamycin) inhibitor. mTOR has been shown to have a significant role in the signaling pathway that regulates cellular metabolism, growth and proliferation and is activated during tumorigenesis.
MTX240 is a molecular glue, bringing two intracellular proteins, PDE3a and SLFN12, specifically co-expressed by GIST cancer cells, into close proximity to form a stable complex. This interaction stabilizes SLFN12, enabling it to drive RNase-mediated apoptosis in GIST cells through a mechanism independent of KIT signaling.
Tolimidone is an orally delivered, potent and selective inhibitor of Lyn kinase. Lyn is a member of the Src family of protein tyrosine kinases, which is mainly expressed in hematopoietic cells, in neural tissues, liver, and adipose tissue. Tolimidone demonstrates glycemic control via insulin sensitization in animal models of diabetes and has the potential to become a first in class blood glucose modulating agent.
Biodexa’s headquarters and R&D facility is in Cardiff, UK. For more information visit www.biodexapharma.com.
Forward-Looking Statements
Certain statements in this announcement may constitute “forward-looking statements” within the meaning of legislation in the United Kingdom and/or United States. Such statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and are based on management’s belief or interpretation. All statements contained in this announcement that do not relate to matters of historical fact should be considered forward-looking statements. In certain cases, forward-looking statements can be identified by the use of words such as “plans”, “expects” or “does not anticipate”, or “believes”, or variations of such words and phrases or statements that certain actions, events or results “may”, “could”, “would”, “might” or “will be taken”, “occur” or “be achieved.” Forward-looking statements and information are subject to various known and unknown risks and uncertainties, many of which are beyond the ability of the Company to control or predict, that may cause their actual results, performance or achievements to be materially different from those expressed or implied thereby, and are developed based on assumptions about such risks, uncertainties and other factors set out herein.
Reference should be made to those documents that Biodexa shall file from time to time or announcements that may be made by Biodexa in accordance with the rules and regulations promulgated by the SEC, which contain and identify other important factors that could cause actual results to differ materially from those contained in any projections or forward-looking statements. These forward-looking statements speak only as of the date of this announcement. All subsequent written and oral forward-looking statements by or concerning Biodexa are expressly qualified in their entirety by the cautionary statements above. Except as may be required under relevant laws in the United States, Biodexa does not undertake any obligation to publicly update or revise any forward-looking statements because of new information, future events or events otherwise arising.
INTRODUCTION
Headquartered in Cardiff, UK, Biodexa is a NASDAQ listed biopharmaceutical company developing innovative products focused on the treatment or prevention of gastrointestinal cancers including Familial Adenomatous Polyposis (“FAP”) and Gastro Intestinal Stromal Tumors (“GIST”).
Until last year, the Company had been developing MTX110 for rare/orphan brain cancers but, due to resource constraints, these programs have been de-prioritized and removed from the R&D pipeline.
STRATEGY
The Company’s transition from a drug delivery company to a higher value therapeutics company was completed in 2025. With the in-licensing of MTX240 from Otsuka Pharmaceutical Co., Limited (“Otsuka”) in February 2026, our pipeline is increasingly focused on gastrointestinal and oncology programs. All of our pipeline of assets are at clinical stage and, we believe, offer significant potential to improve outcomes for patients.
Our priorities for 2026 are as follows:
| Strategic Imperatives | Progress in 2025 | Priorities for 2026 |
| Advance our development assets through the clinic | We announced the following clinical milestones in 2025:
March: appointment of Precision for Medicine as our Contract Research Organization to manage the European component of our Phase 3 Serenta trial March: a successful Type C meeting with FDA, paving the way for the initiation of the Phase 3 Serenta trial in the US June: first patient enrolled in a Phase 2a study of tolimidone in T1D by the University of Alberta Diabetes Institute August: enrolment of the first US patient in the Phase 3 Serenta trial November: receipt of approval of Clinical Trial Application from EMA December: enrolment of the first European patient in the Phase 3 Serenta trial |
Accelerate recruitment of our Phase 3 Serenta trial of eRapa in patients with FAP in the US and Europe and possibly Asia
Continued engagement with FAP patient support groups in the US and Europe Complete a bioavailability study of eRapa compared with Rapamune® Initiate a Phase 1b/2a dose escalation and extension study of MTX240 in GIST |
| Develop and broaden our drug development pipeline | We negotiated the in-license of MTX240 from Otsuka during the latter half of 2025 and signed the Licensing and Collaboration Agreement in early February 2026
The US patent covering “oral rapamycin nanoparticle preparations and use” was issued in February |
Appoint a contract manufacturer to manufacture clinical trial supplies for a Phase 1b/2a study of MTX240 in GIST and initiate a Phase 1b/2a study of MTX240 in GIST
Initiate preclinical experiments to support additional potential, ideally orphan, indications for tolimidone Seek additional pre-IND and/or clinical-stage assets to acquire or in-license Expand further our patent portfolio to cover new inventions and divisionals to strengthen existing patent families |
| Secure long term, ideally non-dilutive financing for the Company | In May we announced our partner, Emtora Biosciences, had successfully secured an additional $3.0 million (bringing the total to $20.0 million) grant funding from the Cancer Prevention and Research Institute of Texas (“CPRIT”)
In December we announced a $10.0 million financing at $3.28 per ADS with two Series L warrants for each ADS |
Secure licensees and/or co-development partners for eRapa in Europe and Japan
Investigate other potential sources of long-term capital to support the Company’s development programs |
BUSINESS MODEL
Having successfully broadened our internal pipeline, our business model is to add value to our development programs by advancing them through the clinic before seeking partners to complete late-stage studies and commercialize the products.
Development
Our intention is to build a balanced portfolio of clinical-stage development assets, ideally with a focus on gastrointestinal and oncology indications. eRapa was in-licensed in April 2024 and is undergoing a registrational Phase 3 trial in FAP together with an ongoing Phase 2 study in NMIBC. MTX240 was in-licensed in February 2026 as Phase 1 ready for GIST. Tolimidone, which was in-licensed in December 2023, is in Phase 2a and is being developed initially for T1D.
Manufacturing
We do not intend to establish our own manufacturing capabilities. For clinical trial supplies we utilize GMP-certified contract manufacturers.
Commercialization
Once proof-of-concept has been established, we intend to out-license our products to a partner who would complete the clinical development and subsequently market and sell them in the licensed territory. In addition to reimbursement of development costs, the partner would be expected to make milestone payments based on sales targets and royalty payments. Because the Phase 3 program for eRapa in FAP is substantially funded by a grant from CPRIT and an escrowed company match, commercialization of eRapa in FAP by the Company remains an option.
Our development pipeline now includes four projects, all which are at clinical stage, as follows:
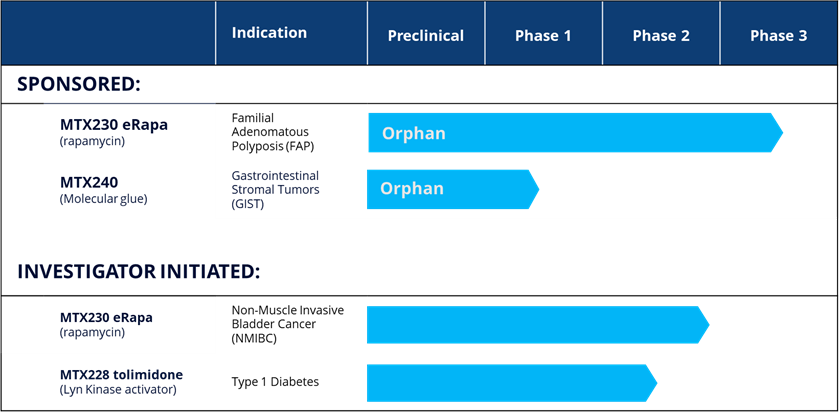
CLINICAL-STAGE ASSETS
eRapa
eRapa is a proprietary oral capsule formulation of rapamycin, also known as sirolimus. Rapamycin is an mTOR (mammalian Target Of Rapamycin) inhibitor. mTOR has been shown to have a significant role in the signaling pathway that regulates cellular metabolism, growth and proliferation and is activated during tumorgenesis1. Rapamycin is approved in the US for organ rejection in renal transplantation as Rapamune® (Pfizer). Through the use of nanotechnology and pH sensitive polymers, eRapa is designed to address the poor bioavailability, variable pharmacokinetics and toxicity generally associated with the currently available forms of rapamycin.
Familial Adenomatous Polyposis (FAP)
FAP is characterized as a proliferation of polyps in the colon and/or rectum, usually occurring in mid-teenage years. There is no approved therapeutic option for treating FAP patients, for whom active surveillance and surgical resection of the colon and/or rectum remain the standard of care. If untreated, FAP typically leads to cancer of the colon and/or rectum. There is a significant hereditary component to FAP with a reported incidence of one in 5,000 to 10,000 in the US2 and one in 11,300 to 37,600 in Europe3. eRapa has received Orphan Designation in the US and Europe. Importantly, mTOR has been shown to be over-activated in FAP polyps, thereby underscoring the rationale for using a potent and safe mTOR inhibitor like eRapa to treat FAP. eRapa received FDA Fast Track designation in February 2025.
The results of the Phase 2 study were presented at two leading scientific conferences in the second quarter of 2024. Following a positive Type C meeting with the FDA, the protocol for a registrational Phase 3 trial was finalized. The Phase 3 trial, branded “Serenta”, is a multi-center, double-blind, placebo-controlled study in high risk patients diagnosed with FAP. The study plans to recruit 168 patients randomized 2:1 drug:placebo across 30 or more sites in the US and Europe, with a primary endpoint being time to a defined progression free survival event. Quality of life measures will also be captured. The study is expected to recruit over 18 months and is supported by a non-dilutive grant of $20.0 million from CPRIT.
Non-muscle Invasive Bladder Cancer (NMIBC)
NMIBC refers to tumors found in the tissue that lines the inner surface of the bladder. The most common treatment is transurethral resection of the bladder tumor followed by intravesical Bacillus Calmette-Guerin (“BCG”) with chemotherapy depending upon assessment of risk of recurrence. NMIBC is the fourth most common cancer in men with an incidence of 10.1 per 100,000 in men and 2.5 per 100,000 in women4. An ongoing double-blind, placebo-controlled Phase 2 Investigator Initiated Trial (“IIT”) in NMIBC is fully enrolled at 166 patients with primary endpoints of safety/tolerability and relapse free survival after 12 months of treatment. The Phase 2 study is supported by a $2.8 million non-dilutive grant from the National Cancer Institute, part of the National Institutes of Health.
MTX240
MTX240, discovered by Otsuka and originally coded OPB-171775, is a novel molecular glue we intend to develop initially for the treatment of GIST. MTX240’s molecular glue activity brings together two intracellular proteins, PDE3A and SLFN12, specifically co-expressed by GIST cancer cells, into close proximity to form a stable complex. This interaction stabilizes SLFN12, enabling it to drive RNase-mediated apoptosis in GIST cells through a mechanism independent of KIT or PDGFR signaling. GIST is mostly driven by activating mutations in the KIT or PDGFR receptor tyrosine kinase. Although tyrosine kinase inhibitors (“TKIs”) such as imatinib, sunitinib, and regorafenib have significantly improved outcomes for GIST patients, resistance almost always develops through secondary KIT or PDGFR mutations or activation of alternative signaling pathways. This represents a substantial clinical challenge with limited therapeutic options for patients once they have cycled through the available TKIs. Molecular glue technology represents a novel approach that induces targeted protein interactions, offering a distinct mechanism of action to conventional tyrosine kinase inhibitors for GIST and by triggering cell death through an alternative MAPKinase pathway. MTX240 is designed to overcome the resistance mechanisms that render TKI-resistant GISTs refractory to conventional TKIs.
Gastrointestinal Stromal Tumors (GIST)
GIST is a rare gastrointestinal malignancy with approximately 6,000 diagnosed patients annually in the US5, with a significant unmet medical need for patients who develop TKI resistance. Approximately 10-15%. of GIST patients are either primarily refractory, or develop secondary resistance to available TKIs whereupon options for these patients remain limited.
The global GIST market is valued at approximately $1.3 billion and is expected to grow at 6-10% annually through 20326, driven by rising incidence and emerging therapeutic options targeting treatment-resistant disease.
GIST qualifies for orphan drug designation in major regulatory jurisdictions, offering potential regulatory advantages and incentives to support drug development.
Tolimidone
Tolimidone was originally discovered by Pfizer Inc. (“Pfizer”) and was developed through Phase 2 for the treatment of gastric ulcers. Pfizer undertook a broad pre-clinical program to characterize the pharmacology, pharmacokinetics, metabolism and toxicology of tolimidone. Pfizer discontinued development of the drug due to lack of efficacy for that indication in Phase 2a. Tolimidone is a selective activator of the enzyme Lyn kinase which increases phosphorylation of insulin substrate -1, thereby amplifying the signaling cascade initiated by the binding of insulin to its receptor.
Type 1 Diabetes (T1D)
In T1D, the body’s immune system attacks pancreatic beta cells such that they can no longer produce insulin which is required to regulate plasma glucose levels. The causes of T1D are not fully understood and there is currently no cure. Patients with T1D are dependent on daily administration of insulin (via injection or infusion).
We are developing tolimidone for the treatment of T1D. As a Lyn kinase activator, tolimidone has been shown in preclinical experiments to have a role in beta cell survival and proliferation. If replicated in clinical studies, tolimidone could have the potential to be disease modifying and change the treatment paradigm for T1D. T1D affects approximately 8.4 million people worldwide and there are approximately 500,000 new diagnoses per annum7.
CHIEF EXECUTIVE’S REVIEW
Introduction
In 2025, our primary focus was the successful initiation of the Phase 3 Serenta trial of eRapa in FAP. Considerable effort was also expended on due diligence and negotiation of the Licensing and Collaboration Agreement for MTX240 which was signed in early February 2026.
R&D update
eRapa
We in-licensed eRapa, a proprietary formulation of rapamycin, from Rapamycin Holdings, Inc. d/b/a Emtora Biosciences, Inc. (“Emtora”) in April 2024. We made considerable progress with our Phase 3 program for eRapa in FAP during 2025 and achieved a number of important milestones as set out below.
In March 2025 we announced the appointment of Precision for Medicine, LLC (“Precision”) as the CRO to manage the European component of our Phase 3 trial. With over 20 years of experience, Precision’s reputation is founded on its high-caliber, therapeutically specialized staff, experienced scientists and physicians, advanced specialty laboratories, and problem-solving capabilities. Precision has conducted 333 clinical trials in rare diseases and employs over 700 team members in Europe across 11 locations.
Also, in March 2025 we reported that we had a productive Type C meeting with the FDA which included a discussion of the statistical plan, the safety database and, most importantly, a composite endpoint for the Phase 3 Serenta trial. FDA representatives from both Gastroenterology and Oncology Divisions provided valuable input into the proposed program, providing a clear path forward for finalization of the protocol.
Following initiation of the first US clinical site in June 2025, the first US patient was enrolled in August 2025. In November 2025 we received approval from the EMA for our Clinical Trial Application for the Phase 3 trial and the first European patient was enrolled in December 2025. As of 16 March, there were 22 active clinical sites and 37 patients enrolled in the Serenta study.
Other milestones achieved in 2025 included the receipt of Fast Track designation from FDA and Orphan Drug Designation from the EMA.
The Phase 3 study is expected to take approximately 18 months to recruit all 168 patients and database lock will occur after 75 events which is expected to be in the second half of 2029.
An ongoing double-blind, placebo-controlled Phase 2 IIT of eRapa in NMIBC has enrolled all 166 patients and is expected to read out in the third quarter of 2026.
MTX240
In February 2026 we announced that we had entered into a License and Collaboration Agreement with Otsuka for MTX240. Under the terms of the license agreement we have exclusive rights to develop and commercialize MTX240 globally with the exception of Japan where Otsuka retains its rights. The agreement included a modest upfront fee and provides for one additional development milestone and low double-digit approval milestones. In addition, tiered royalties in the mid-single digit range are payable on net sales of MTX240.
As a molecular glue, MTX240 has a unique mechanism of action compared with tyrosine kinase inhibitors, the current standard of care for GIST, all of which can eventually lose efficacy through continued mutation. MTX240’s novel mechanism may provide clinical benefit for a significant proportion of GIST patients, not only those who have developed resistance to TKIs. In patient derived xenograft (PDX) models, MTX240 has shown dose-dependent anti-tumor efficacy in imatinib and sunitinib resistant models irrespective of KIT mutation status.
MTX240 benefits from composition of matter patents in the US, Europe, Japan and various other countries extending through 2037 excluding any patent term extension.
Our first step is to manufacture clinical trial supplies of MTX240 and then initiate an open-label Phase 1b/2a study by year-end. The study is expected to be in two parts: a standard dose escalation part to determine a maximum tolerated dose followed by an extension part. The extension part is likely to enroll patients with TKI-resistant GIST. By focusing on this high-need population, we are aiming to rapidly validate MTX240’s potential to treat patients who do not, or no longer, respond to the current standard of care.
Tolimidone
In December 2023 we secured the global rights to develop and commercialize tolimidone. The product is supported by very substantial preclinical data, has been exposed to more than 700 patients and has demonstrated compelling preclinical data to support our chosen indication of T1D. As a first step in the planned continued clinical development of tolimidone, we initiated a Phase 2a dose confirmation IIT at the University of Alberta, Diabetes Institute with the first patient enrolled in June 2025. The Phase 2a study is open-label and is expected to enroll approximately 15 patients with T1D treated with tolimidone for three months with endpoints of change in C-peptide levels, HbA1c and number of hyperglycemic events.
MTX110
Due to resource constraints, the MTX110 rare/orphan brain cancer programs were not funded in 2025 and, accordingly, have been removed from our development pipeline.
Financings
Promissory Note
In December 2024 we issued an unsecured promissory note to C/M Capital Master Fund LP in the principal amount of $600,000 with a 10% original issue discount. The Note was an unsecured obligation with interest of 5% pa. and was fully repaid in December 2025.
Equity Line of Credit (ELOC)
In January 2025, we entered into a three-year $35.0 million ELOC with C/M Capital Master Fund, LP. During calendar 2025 we used the ELOC to raise approximately $8.9 million in gross proceeds.
CPRIT grant
In May 2025 we announced CPRIT had awarded an additional grant of $3.0 million, bringing the total to $20.0 million, to support the Phase 3 program of eRapa in FAP. Receipt of funds is conditional upon a 1:1 company match.
Registered Offering
In December 2025 we closed a registered offering of ADSs and pre-funded warrants at $3.28 per ADS to raise gross proceeds of $10.0 million. We also issued 6.10 million Series L warrants exercisable at $3.28 over five years.
Further details of financings are included in “Financial Review”.
Outlook
Our primary focus for 2026 will be two-fold: first, continued acceleration of enrolment of patients in our registrational Phase 3 Serenta trial in the US and Europe and by adding additional countries and sites and second, manufacture of clinical supplies and initiation of a Phase 1b/2a study of MTX240 in GIST.
As has been the case for the past few years, financing for small- and micro-cap biotech companies remains challenging. The $10.0 million registered offering in December 2025 extends our runway into 3Q26 and the $35.0 million ELOC we put in place in February 2025 provides a potential backstop source of capital.
1 Tian et al., mTOR Signaling in Cancer and mTOR Inhibitors in Solid Tumor Targeting Therapy, Int J Mol Sci. 2019 Feb; 20(3): 755
2 www.rarediseases.org
3. www.orpha.net
- Cassell et al., World J Oncol. 2019 Jun; 10(3): 123–131
5 Zhu, H., et al. (2023). Update of epidemiology, survival and initial treatment in gastrointestinal stromal tumor: A population-based analysis. BMJ Open, 13(7), e072945. https://doi.org/10.1136/bmjopen-2023-072945
6 DataBridge Market Research. (2023). Gastrointestinal stromal tumor market size, trends and forecasts (2024-2032). Retrieved from https://www.databridgemarketresearch.com/reports/global-gastrointestinal-stromal-tumor-market
7 Medicina Apr 2023
FINANCIAL REVIEW
Introduction
Biodexa Pharmaceuticals PLC was incorporated as a company on 12 September 2014 and is domiciled in England and Wales.
Financial analysis
Key performance indicators
| 2025 | 2024 | Change | |
| R&D expenditure | £3.96m | £5.44m | (27.2)% |
| R&D as % of operating costs | 45% | 59% | n/a |
| Net cash inflow/(outflow) for the year | £6.87m | (£4.30m) | n/m |
| ============ | ============ | ============ | |
Revenue
Revenue for both periods was £Nil. The last of the Company’s R&D collaborations concluded in September 2023.
Research and development expenditure
R&D costs analyzed by development project indication were as follows:
| Year ended 31 December | 2025 | 2024 | |
| £’000 | £’000 | ||
| MTX230 eRapa | |||
| Familial Adenomatous Polyposis (net of grant) | 1,338 | 1,627 | |
| Non-muscle Invasive Bladder Cancer | 154 | 284 | |
| Total eRapa | 1,492 | 1,911 | |
| MTX228 Tolimidone | |||
| Type 1 Diabetes | 383 | 1,093 | |
| Total tolimidone | 383 | 1,093 | |
| MTX110 (panobinostat) | |||
| Diffuse Midline Glioma | – | (6) | |
| Recurrent Glioblastoma | 36 | 635 | |
| Medulloblastoma | – | – | |
| Total MTX110 (panobinostat) | 36 | 629 | |
| Other preclinical | – | 102 | |
| R&D overheads | 2,050 | 1,702 | |
| Total R&D | 3,961 | 5,437 | |
MTX230 eRapa Familial Adenomatous Polyposis costs are shown above net of grant income. This is analyzed as follows:
| Year ended 31 December | 2025 | 2024 | |
| £’000 | £’000 | ||
| Grant income | (4,458) | (1,215) | |
| Gross costs | 5,796 | 2,842 | |
| Net charge to income statement | 1,338 | 1,627 | |
| % costs allocated against CPRIT grant | 77% | 43% | |
Research and development costs were £3.96 million, a reduction of £1.48 million, or 27% on 2024 (2024: £5.44 million). The percentage of R&D costs as a percentage of operating costs reduced to 45% from 59% in the prior year. The reduction in R&D costs in 2025 predominately reflects a reduction in spend of £0.71 million on pre-clinical studies and manufacturing costs on MTX228, £0.60 million on the MTX110 MAGIC-G1 study in rGBM and £0.44 million on MTX230 eRapa (net of CPRIT grant income). The percentage of MTX230 (eRapa) costs that were able to be offset against grant funding during the period was 77% compared to 43% in 2024. We anticipate that over the life of the grant this will be 67%.
Administrative costs
Administrative costs in the year increased by £1.04 million to £4.84 million (2024: £3.79 million), an increase of 27%. The increase in administrative costs in the year is driven by foreign exchange movement of £0.44 million, an increase in professional fees of £0.73 million offset by a reduction in share-based payments of £0.10 million.
In 2025 the Company expensed £1.72 million on legal and professional fees in connection with the successful financing transaction in December, the acquisition of the Otsuka license completed in 2026 and aborted acquisitions, and £0.37 million non- cash upfront commitment fee for the ELOC, this compares to £0.88m spent in 2024 on similar transactions.
Staff costs
During the year, the average number of staff decreased to 11 (2024: 13). Total staff cost increased 1% to £2.17 million (2024: £2.15 million), driven by the increase in wages and salaries of £0.14 million.
Finance income and expense
Finance income in 2025 and 2024 included gains in respect of an equity settled derivative financial liability of £2.30 million (2024: £3.22 million). The gains arose as a result of the fall in the Biodexa share price. In addition, the Company earned interest on cash deposits.
Finance expense in the 2024 and 2025 related to lease liabilities, discounted interest on deferred consideration and interest on the promissory note issued in December 2024.
Taxation
During 2025 and 2024 we recognized U.K research and development tax credits of £0.11 million and £0.25million in respect of R&D expenditure incurred.
Capital expenditure
Purchase of tangible fixed assets in 2025 was £2,000 (2024: £0.01 million) and related to the purchase of IT equipment. During the year the Company took the decision to de-commission its laboratory in Cardiff and disposed of the majority of its equipment. This is in line with the Company strategy as set out above.
Cash flow
Net cash outflow from operating activities in 2025 was £5.59 million (2024: outflow £12.26 million) driven by a net loss of £6.38 million (2024: loss £5.73 million) and after positive movements in working capital of £1.72 million (2024: negative £3.74 million), taxes received of £0.71 million (2024: £0.13 million), and other net negative adjustments for non-cash items totaling £1.63 million (2024: negative £2.93 million).
Investing activities outflow in 2025 of £0.58 million (2024: outflow of £0.60 million) included purchases of property, plant and equipment of £2,000 (2024: £0.01 million) and payment of deferred consideration on the eRapa license of £0.69 million (2024: £0.77 million). These cash outflows are offset by interest income from bank deposits of £0.09 million (2024: £0.18 million).
Financing activities inflow in 2025 of £13.04 million (2024: inflow of £8.56 million) was driven by receipts from share issues of £13.75 million (2024: £8.31 million) from use of the ELOC agreement, proceeds from the warrant inducement in May 2025 and proceeds from the registered offering in December 2025. This is offset by loan repayments (including interest) of £0.46 million and payments on lease liabilities of £0.25 million (2024: £0.19 million).
As a result of the foregoing, net cash inflow for the year was £6.87 million (2024: outflow of £4.30 million).
Change in ADS Ratio and Nominal Value of Ordinary Shares
On 31 July 2025 the Company effected a change in the ratio of the Company’s Ordinary Shares from each ADS representing 10,000 Ordinary Shares to each ADS representing 100,000 Ordinary Shares.
At a General Meeting on 11 June 2025, shareholders approved the subdivision and redesignation of the Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless.
On 18 March 2026 the Company announced a change in the ratio of the Company’s Ordinary Shares from each ADS representing 100,000 Ordinary Shares to each ADS representing 500,000 Ordinary Shares.
Financings
Equity Line of Credit (“ELOC”)
In January 2025, we entered into a securities purchase agreement, or equity line of credit (“ELOC”), with the newly formed C/M. Under the terms of the ELOC, we have the right, but not the obligation, to sell to C/M from time to time up to $35 million of newly issued ADSs over a 36-month period, unless the ELOC is terminated. As consideration for the execution and delivery of the ELOC, we agreed to pay a commitment fee (“Commitment Fee”) of $875,000 in cash, of which (i) $612,500 was to be paid to C/M on signing the ELOC and (ii) the balance was to be paid pro rata, simultaneously with the delivery of any ADSs sold under the ELOC. We had the right to issue ADSs representing the value of the applicable portion of the Commitment Fee. We paid the initial Commitment Fee of $612,500 through the issuance of 140,080 Depositary Shares to the Purchaser.
We may direct C/M to purchase a specified number of ADSs not to exceed $2.5 million on any given day, at a price based on a formula, typically 95% of the closing price on the prior day. As of 31 December 2025, the Company had raised gross proceeds of $8.92 million from the ELOC.
Warrant Inducement
In May 2025 we entered into letter agreements with certain holders of outstanding Series E, Series H, Series J and Series K warrants to reduce the exercise price of such warrants to $0.31 per share. The holders exercised an aggregate of 200,433 warrants representing the same number of ADSs. We received gross proceeds of approximately $62,000, before offering expenses. The Company did not issue new warrants to replace the exercised warrants and did not engage a placement agent to facilitate the transaction.
Registered Offering
In December 2025 we closed a Registered Offering with institutional investors for the sale of (i) an aggregate of 157,000 ADSs and (ii) an aggregate of 2,891,781 pre-funded warrants exercisable for Depositary Shares at a price per Depositary Share of $3.28 and a price per pre-funded warrant of $3.2799. The pre-funded warrants were exercisable immediately. In addition, we also issued and sold to investors Series L Warrants for 6,097,562 Depositary Shares at an exercise price of $3.28 per Depositary Share. The Series L Warrants are exercisable immediately and expire five years from the date of issuance. We received gross proceeds of $10 million before deducting placement agent’s fees and related offering expenses.
Promissory Note
The promissory note issued in December 2024 for $600,000 was fully repaid during the year.
CPRIT Grant
In May 2025 we announced that our collaboration partner, Emtora, had been awarded an additional grant of $3.0 million from CPRIT. The award brought the total grant awarded by CPRIT to support the registrational Phase 3 program of eRapa in FAP to $20.0 million. Access to the $3.0 million is conditioned on the Company providing a 1:1 match of $3.0 million.
Going Concern – material uncertainty
The Group and Company has experienced net losses and significant cash outflows from cash used in operating activities over the past years as it develops its portfolio. For the year ended 31 December 2025, the Group incurred a consolidated loss for the year of £6.38 million and negative cash flows from operations of £5.59 million. As of 31 December, 2025, the Group had an accumulated deficit of £155.81 million.
The Group’s future viability is dependent on its ability to raise cash from financing activities to finance its development plans until milestones and/or royalties can be secured from partnering the Company’s assets. The Group’s failure to raise capital as and when needed could have a negative impact on its financial condition and ability to pursue its business strategies.
The Directors believe there are adequate options and time available to secure additional financing for the Company and after considering the uncertainties, the Directors consider it is appropriate to continue to adopt the going concern basis in preparing these financial statements. The Group’s consolidated financial statements have therefore been presented on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business.
As at 31 December 2025, the Group had cash and cash equivalents of £8.53 million. The Directors have prepared cash flow forecasts and considered the cash flow requirement for the Group for the next three years including the period 12 months from the date of approval of the consolidated financial statements. These forecasts show that further financing will be required in Q3 2026 assuming, inter alia, that certain development programs and other operating activities continue as currently planned. Pursuant to its $35 million Equity Line of Credit, or ELOC, as described above, the Company may direct C/M to purchase ADSs (subject to certain limitations) and receive proceeds in accordance with a formula price. There is no guarantee that the Company will be able to use the ELOC or rase from other financing to the extent necessary to finance the Company’s operations. As at 31 December 2025 $26.08 million remains undrawn from the ELOC.
In the Directors’ opinion, the environment for financing of small and micro-cap biotech companies remains challenging. While this may present acquisition and/or merger opportunities with other companies with limited or no access to financing, as noted above, any attendant financings by Biodexa are likely to be dilutive. The Directors continue to evaluate financing options, including those connected to acquisitions and/or mergers, potentially available to the Group. Any alternatives considered are contingent upon the agreement of counterparties and accordingly, there can be no assurance that any alternative courses of action to finance the Company would be successful.
This requirement for additional financing in the short term represents a material uncertainty that may cast significant doubt upon the Group and Parent Company’s ability to continue as a going concern. Should it become evident in the future that there are no realistic financing options available to the Company which are actionable before its cash resources run out then the Company will no longer be a going concern. In such circumstances, we would no longer be able to prepare financial statements under paragraph 25 of IAS 1. Instead, the financial statements would be prepared on a liquidation basis and assets would be stated at net realizable value and all liabilities would be accelerated to current liabilities.
Environmental matters, community, human rights issues and employees
As at 31 December 2025 the Group had 11 employees, of whom 4 were routinely based at its offices in Cardiff, Accordingly the Company believes it has a relatively modest environmental impact. A number of policies and procedures governing expectations of ethical standards and the treatment of employees and other stakeholders are set out in the Company’s Employee Handbook. The Company has also established an anti-slavery policy pursuant to the Modern Slavery Act 2015.
The Company strives to be an equal opportunity employer, irrespective of race or gender. At 31 December 2025, the number of male/female employees was 36%/64%, the number of male/female senior managers was 57%/43% and the number of male/female Directors was 80%/20%.
Annual greenhouse gas emissions
We measure our environmental performance by reporting our carbon footprint in terms of tonne CO2 equivalent. We report separately on our indirect emissions from consumption of electricity (Scope 2) and emissions consisting of employee travel in cars on Group business estimated on the basis of miles travelled (Scope 3). The Group have elected to monitor and report its energy efficiency using tonnes of CO2 per employee as an intensity ratio.
Methodology
In calculating the reported energy usage and equivalent greenhouse gas emissions the Group have referred to the HM Government Environment Reporting Guidelines and the GHG Reporting Protocol. A location-based allocation methodology was used to calculate electricity usage.
| Tonnes CO2e | 2025 | 2024 |
| Scope 2 | 10 | 17 |
| Scope 3 | 1 | 3 |
| Total | 12 | 20 |
| Intensity ratio (tonnes of CO2 per employee) | 1.1 | 0.9 |
The Group’s electricity costs for 2025 were approximately £16,000 (2024: £25,000). The kWh usage in the year was 54,380 (2024: 81,933). The Group has no immediate plans to improve energy efficiency.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME
For the year ended 31 December
| Note | 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Revenue | – | – | 381 | |
| Other income | 152 | 31 | 14 | |
| Research and development costs | (3,961) | (5,437) | (4,067) | |
| Administrative costs | (4,836) | (3,793) | (4,342) | |
| Loss from operations | (8,645) | (9,199) | (8,014) | |
| Finance income | 2 | 2,385 | 3,385 | 570 |
| Finance expense | 2 | (229) | (165) | (41) |
| Loss before tax | (6,489) | (5,979) | (7,485) | |
| Taxation | 105 | 250 | 406 | |
| Loss for the year attributable to the owners of the parent | (6,384) | (5,729) | (7,079) | |
| Other comprehensive income: | ||||
| Items that will or may be reclassified subsequently to profit or loss: | ||||
| Exchange gains arising on translation of foreign operations | 3 | – | – | |
| Total other comprehensive income net of tax | 3 | – | – | |
| Total comprehensive loss attributable to the owners of the parent | (6,381) | (5,729) | (7,079) | |
| Loss per share | ||||
| Continuing operations | ||||
| Basic and diluted loss per ordinary share – pence | 3 | (0.01)p | (0.1)p | (2)p |
The notes form an integral part of these consolidated financial statements.
CONSOLIDATED STATEMENTS OF FINANCIAL POSITION
At 31 December
| Company number 09216368 | Note | 2025 £’000 |
2024 £’000 |
2023 £’000 |
| Assets | ||||
| Non-current assets | ||||
| Property, plant and equipment | 91 | 324 | 571 | |
| Intangible assets | 5,645 | 5,646 | 2,941 | |
| 5,736 | 5,970 | 3,512 | ||
| Current assets | ||||
| Trade and other receivables | 3,786 | 6,568 | 637 | |
| Current taxation receivable | 123 | 573 | 422 | |
| Cash and cash equivalents | 8,534 | 1,669 | 5,971 | |
| 12,443 | 8,810 | 7,030 | ||
| Total assets | 18,179 | 14,780 | 10,542 | |
| Liabilities | ||||
| Non-current liabilities | ||||
| Deferred consideration | 645 | 1,306 | – | |
| Borrowings | – | 118 | 295 | |
| 645 | 1,424 | 295 | ||
| Current liabilities | ||||
| Trade and other payables | 2,590 | 3,504 | 1,240 | |
| Deferred consideration | 563 | 538 | – | |
| Borrowings | 61 | 609 | 169 | |
| Provisions | – | – | – | |
| Derivative financial liability | 2,915 | 383 | 4,160 | |
| 6,129 | 5,034 | 5,569 | ||
| Total liabilities | 6,774 | 6,458 | 5,864 |
| CONSOLIDATED STATEMENTS OF FINANCIAL POSITION(CONTINUED)
At 31 December |
||||
| Note | 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Issued capital and reserves attributable to owners of the parent | ||||
| Share capital | 14,099 | 11,725 | 6,253 | |
| Share premium | 98,923 | 93,124 | 86,732 | |
| Merger reserve | 53,003 | 53,003 | 53,003 | |
| Warrant reserve | 1,185 | 894 | 3,457 | |
| Foreign exchange reserve | 3 | – | – | |
| Accumulated deficit | (155,808) | (150,424) | (144,767) | |
| Total equity | 11,405 | 8,322 | 4,678 | |
| Total equity and liabilities | 18,179 | 14,780 | 10,542 | |
The notes form an integral part of these consolidated financial statements.
CONSOLIDATED STATEMENTS OF CASH FLOWS
For the year ended 31 December
| Note | 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Cash flows from operating activities | ||||
| Loss for the year | (6,384) | (5,729) | (7,079) | |
| Adjustments for: | ||||
| Depreciation of property, plant and equipment | 52 | 117 | 143 | |
| Depreciation of right of use asset | 136 | 135 | 137 | |
| Amortisation of intangible fixed assets | 1 | 2 | 3 | |
| Loss on disposal of property, plant and equipment | 29 | 4 | 2 | |
| Impairment of loan | – | – | 79 | |
| Impairment of ELOC upfront fee | 373 | – | – | |
| Finance income | (2,385) | (3,385) | (570) | |
| Finance expense | 229 | 165 | 41 | |
| Share-based payment charge | 170 | 283 | 28 | |
| Taxation | (105) | (250) | (406) | |
| Foreign exchange losses/(gains) | (130) | 4 | – | |
| Cash flows from operating activities before changes in working capital | (8,014) | (8,654) | (7,622) | |
| Decrease/(Increase) in trade and other receivables | 2,629 | (5,975) | 365 | |
| (Decrease)/Increase in trade and other payables | (913) | 2,239 | (207) | |
| (Decrease)/Increase in provisions | – | – | (207) | |
| Cash used in operations | (6,298) | (12,390) | (7,671) | |
| Taxes received | 707 | 129 | 845 | |
| Net cash used in operating activities | (5,591) | (12,261) | (6,826) |
CONSOLIDATED STATEMENTS OF CASH FLOWS(CONTINUED)
For the year ended 31 December
| Note | 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Investing activities | ||||
| Purchases of property, plant and equipment | (2) | (9) | (26) | |
| Proceeds from disposal of fixed assets | 18 | – | 4 | |
| Purchase of intangible asset/deferred consideration | (689) | (765) | (237) | |
| Loan granted | – | – | (79) | |
| Interest received | 90 | 176 | 73 | |
| Net cash (used in)/generated from investing activities | (583) | (598) | (265) | |
| Financing activities | ||||
| Interest paid | (14) | – | (13) | |
| Amounts paid on lease liabilities | (246) | (186) | (188) | |
| (Repayment)/Proceeds from promissory note | (450) | 431 | – | |
| Share issues including warrants, net of costs | 13,749 | 8,312 | 10,427 | |
| Net cash generated from financing activities | 13,039 | 8,557 | 10,226 | |
| Net increase/(decrease) in cash and cash equivalents | 6,865 | (4,302) | 3,135 | |
| Cash and cash equivalents at beginning of year | 1,669 | 5,971 | 2,836 | |
| Exchange (losses)/gains on cash and cash equivalents | – | – | – | |
| Cash and cash equivalents at end of year | 8,534 | 1,669 | 5,971 |
The notes form an integral part of these consolidated financial statements.
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
For the year ended 31 December
| Note | Share capital £’000 |
Share premium £’000 |
Merger reserve £’000 |
Warrant reserve £’000 |
Foreign exchange reserve £’000 | Accumulated deficit £’000 |
Total equity £’000 |
|
| At 1 January 2025 | 11,725 | 93,124 | 53,003 | 894 | – | (150,424) | 8,322 | |
| Loss for the year | – | – | – | – | – | (6,384) | (6,384) | |
| Foreign exchange translation | – | – | – | – | 3 | – | 3 | |
| Total comprehensive loss | – | – | – | – | 3 | (6,384) | (6,381) | |
| Transactions with owners | ||||||||
| Shares issued under ELOC agreement | 2,030 | 4,817 | – | – | – | – | 6,847 | |
| Costs associated with ELOC agreement | 86 | (77) | – | – | – | – | 9 | |
| Shares issued on 15 May 2025 | 100 | 143 | – | – | – | – | 243 | |
| Costs associated with share issue on 15 May 2025 | – | (8) | – | – | – | – | (8) | |
| Shares issued 19 December 2025 | 158 | 1,110 | – | 1,184 | – | – | 2,452 | |
| Costs associated with share issue on 19 December 2025 | – | (186) | – | (173) | – | – | (359) | |
| Lapse warrants | – | – | – | (720) | – | 720 | – | |
| Share-based payment charge | – | – | – | – | – | 280 | 280 | |
| Total contribution by and distributions to owners | 2,374 | 5,799 | 291 | 1,000 | 9,464 | |||
| At 31 December 2025 | 14,099 | 98,923 | 53,003 | 1,185 | 3 | (155,674) | 11,405 |
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY(CONTINUED)
| Note | Share capital £’000 |
Share premium £’000 |
Merger reserve £’000 |
Warrant reserve £’000 |
Accumulated deficit £’000 |
Total equity £’000 |
|
| At 1 January 2024 | 6,253 | 86,732 | 53,003 | 3,457 | (144,767) | 4,678 | |
| Loss for the year | – | – | – | – | (5,729) | (5,729) | |
| Total comprehensive loss | – | – | – | – | (5,729) | (5,729) | |
| Transactions with owners | |||||||
| Shares issued on 22 May 2024 | 1,614 | 5,048 | – | – | – | 6,662 | |
| Costs associated with share issue on 22 May 2024 | – | (487) | – | – | – | (487) | |
| Shares issued on 22 July 2024 | 2,105 | 79 | – | 2 | – | 2,186 | |
| Costs associated with share issue on 22 July 2024 | – | (55) | – | – | (297) | (352) | |
| Exercise of warrants during the year | 1,602 | 1,739 | – | (2,565) | – | 776 | |
| Issue of shares to purchase intangible asset | 151 | 68 | – | – | – | 219 | |
| Share-based payment charge | – | – | – | – | 369 | 369 | |
| Total contribution by and distributions to owners | 5,472 | 6,392 | – | (2,563) | 72 | 9,373 | |
| At 31 December 2024 | 11,725 | 93,124 | 53,003 | 894 | (150,424) | 8,322 |
NOTES FORMING PART OF THE FINANCIAL STATEMENTS
For the year ended 31 December 2025
1. Basis of preparation
The consolidated financial statements have been prepared in accordance with international accounting standards in conformity with the requirements of the Companies Act 2006, and they are prepared in accordance with international financial reporting standards. The consolidated financial statements have been prepared on a historical cost basis except that the following assets and liabilities are stated at their fair value: certain financial assets and financial liabilities measured at fair value, and liabilities for cash-settled share-based payments.
The financial information contained in this final announcement does not constitute statutory financial statements as defined in Section 435 of the Companies Act 2006. The financial information has been extracted from the financial statements for the year ended 31 December 2025 which have been approved by the Board of Directors, and the comparative figures for the year ended 31 December 2024 and 31 December 2023 are based on the financial statements for that year.
The financial statements for 2024 and 2023 have been delivered to the Registrar of Companies and the 2025 financial statements will be delivered after the Annual General Meeting.
The auditor’s report for the Company’s 2025 Annual Report and Accounts was unqualified but did draw attention to the material uncertainty relating to going concern. The auditor’s report did not contain statements under s498(2) or (3) of the Companies Act 2006.
Whilst the financial information included in this results announcement has been prepared in accordance with International Financial Reporting Standards (IFRSs) this announcement does not itself contain sufficient information to comply with IFRSs. The information in this results announcement was approved by the board on 27 March 2026.
Going concern – material uncertainty
The Group and Company has experienced net losses and significant cash outflows from cash used in operating activities over the past years as it develops its portfolio. For the year ended 31 December 2025, the Group incurred a consolidated loss for the year of £6.38 million and negative cash flows from operations of £5.59 million. As of 31 December 2025, the Group had an accumulated deficit of £155.81 million.
The Group’s future viability is dependent on its ability to raise cash from financing activities to finance its development plans until milestones and/or royalties can be secured from partnering the Company’s assets. The Group’s failure to raise capital as and when needed could have a negative impact on its financial condition and ability to pursue its business strategies.
The Directors believe there are adequate options and time available to secure additional financing for the Company and after considering the uncertainties, the Directors consider it is appropriate to continue to adopt the going concern basis in preparing these financial statements. The Group’s consolidated financial statements have therefore been presented on a going concern basis, which contemplates the realisation of assets and the satisfaction of liabilities in the normal course of business.
As at 31 December 2025, the Group had cash and cash equivalents of £8.53 million. The Directors have prepared cash flow forecasts and considered the cash flow requirement for the Group for the next three years including the period 12 months from the date of approval of the consolidated financial statements. These forecasts show that further financing will be required in Q3 2026 assuming, inter alia, that certain development programs and other operating activities continue as currently planned. Pursuant to its $35 million Equity Line of Credit, or ELOC, as described in the Finance Review, the Company may direct C/M to purchase ADSs (subject to certain limitations) and receive proceeds in accordance with a formula price There is no guarantee that the Company will be able to use the ELOC or raise from other financing to the extent necessary to finance the Company’s operations. As at 31 December 2025 $26.08 million remains undrawn from the ELOC.
In the Directors’ opinion, the environment for financing of small and micro-cap biotech companies remains challenging. While this may present acquisition and/or merger opportunities with other companies with limited or no access to financing, as noted above, any attendant financings by Biodexa are likely to be dilutive. The Directors continue to evaluate financing options, including those connected to acquisitions and/or mergers, potentially available to the Group. Any alternatives considered are contingent upon the agreement of counterparties and accordingly, there can be no assurance that any alternative courses of action to finance the Company would be successful.
This requirement for additional financing in the short term represents a material uncertainty that may cast significant doubt upon the Group and Parent Company’s ability to continue as a going concern. Should it become evident in the future that there are no realistic financing options available to the Company which are actionable before its cash resources run out then the Company will no longer be a going concern. In such circumstances, we would no longer be able to prepare financial statements under paragraph 25 of IAS 1. Instead, the financial statements would be prepared on a liquidation basis and assets would be stated at net realizable value and all liabilities would be accelerated to current liabilities.
Accounting for rRapa and CPRIT grant
On 25 April 2024 the Company entered into a License and Collaboration Agreement (LCA) with Rapamycin Holdings, Inc. (d/b/a Emtora Biosciences). The LCA entered into with Emtora meets the definition of a Joint Arrangement under IFRS 11, specifically related to the FAP program.
A jointly controlled escrow account was established on completion of the LCA. FAP program transactions eligible to be allocated against the CPRIT grant and match funding are processed through the escrow account, including the Company’s deposits of matching funds, as set out in the agreement, the receipt of grant funding from CPRIT and the payment of eligible R&D expenses. Although the CPRIT grant and R&D supplier contracts are with Emtora, the joint arrangement nature of the LCA results in Emtora being deemed to be acting as the Company’s agent. Accordingly, the Company recognises 100% of the grant and 100% of the R&D expenditure. The CPRIT grant recognised is on a 1 for 2 match basis for the first $17 million, the final $3 million is recognised on a 1 for 1 basis. In accordance with the Company’s accounting policy, the grant, as it is the re-imbursement of directly related costs, is credited to R&D costs in the same period in the Statements of Comprehensive Income. The escrow account is recognised within prepayments, CPRIT grant received in advance is recognised within deferred revenue and any grant not yet received is recognised in accrued income.
The balances as at 31 December were as follows in relation to the FAP project:
| 31 December 2025
£’000 |
31 December 2024
£’000 |
|
| Prepayments* | 2,842 | 6,114 |
| Deferred revenue | 571 | 1,468 |
* prepayment reflects only the escrow account balance
2. Finance income and expense
| 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Finance income | |||
| Interest received on bank deposits | 83 | 166 | 73 |
| Other interest receivable | 6 | 1 | 10 |
| Gain on equity settled derivative financial liability | 2,296 | 3,218 | 487 |
| Total finance income | 2,385 | 3,385 | 570 |
| 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Finance expense | |||
| Interest expense on lease liabilities | 10 | 19 | 28 |
| Interest expense on deferred consideration | 159 | 144 | – |
| Other loans | 60 | 2 | 13 |
| Total finance expense | 229 | 165 | 41 |
3. Loss per share
| 2025 £’000 |
2024 £’000 |
2023 £’000 |
|
| Numerator | |||
| Loss used in basic EPS and diluted EPS: | |||
| Continuing operations | (6,384) | (5,729) | (7,079) |
| Denominator | |||
| Weighted average number of ordinary shares used in basic EPS: | 54,861,066,264 | 4,952,784,179 | 315,849,600 |
| Basic and diluted loss per share: | |||
| Continuing operations – pence | (0.01)p | (0.1)p | (2)p |
At a General Meeting on 1 June 2025, shareholders approved the subdivision and redesignation of the Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless. The share sub-division and redesignation did not impact the calculation of the denominator as the number of Issued Ordinary Shares did not change.
During the year the Company issued warrants that were accounted through the Warrant Reserve.
The Company has considered the guidance set out in IAS 33 in calculating the denominator in connection with the issuance of Pre-Funded warrants. Management have recognised the warrants from the date of grant rather than the date of issue of the corresponding Ordinary Shares when calculating the denominator.
The Group has made a loss in the current and previous periods presented, and therefore the options and warrants are anti-dilutive. As a result, diluted loss per share is presented on the same basis as basic loss per share.
4. Share capital
| Authorised, allotted and fully paid – classified as equity |
2025 Number |
2025 £ |
2024 Number |
2024 £ |
2023 Number |
2023 £ |
| At 31 December | ||||||
| Ordinary shares of £0.000001 each |
225,817,808,922 | 225,818 | 6,685,918,922 | 334,296 | 1,189,577,722 | 1,189,578 |
| ‘A’ Deferred shares of £1 each | 1,000,001 | 1,000,001 | 1,000,001 | 1,000,001 | 1,000,001 | 1,000,001 |
| ‘B’ Deferred shares of £0.001 each | 4,063,321,418 | 4,063,321 | 4,063,321,418 | 4,063,321 | 4,063,321,418 | 4,063,321 |
| ‘C’ Deferred shares of £0.00005 each | 126,547,389,518 | 6,327,370 | 126,547,389,518 | 6,327,370 | – | – |
| ‘D’ Deferred shares of £0.000001 each | 2,482,747,137,178 | 2,482,747 | – | – | – | – |
| Total | 14,099,257 | 11,724,988 | 6,252,900 |
At a General Meeting on 11 June 2025, shareholders approved the subdivision and redesignation of the Company’s Issued Ordinary Shares of £0.00005 each into to one Ordinary Share of £0.000001 each and 49 ‘D’ Deferred Shares of £0.000001 each. The ‘D’ Deferred Shares have limited rights and are effectively valueless.
On 31 July 2025 the Company effected a ratio change in the number of Ordinary Shares represented by ADSs from 10,000 Ordinary Shares per ADS to 100,000 Ordinary Shares per ADS.
In accordance with the Articles of Association for the Company adopted on 11 June 2025, the share capital of the Company consists of an unlimited number of ordinary shares of nominal value £0.000001 each. Ordinary and deferred shares were recorded as equity.
5. Related party transactions
The Directors consider there to be no related party transactions during the periods reported other than Directors Remuneration.
6. Events after the reporting date
On February 4, 2026, the Company announced that it had entered into a license and collaboration agreement (the “License Agreement”) with Otsuka Pharmaceutical Co., Ltd. (“Otsuka”), pursuant to which Otsuka granted the Company an exclusive, worldwide (excluding Japan) license (the “Licensed Territory”) to develop manufacture and commercialize OPB-171755, to be designated MTX240 (“MTX240”), a Phase 1-ready molecular glue therapeutic candidate, for all human therapeutic uses. The Company intends to initially develop MTX240 for the treatment of gastrointestinal stromal tumors.
Pursuant to the License Agreement, the Company will be responsible for all development, manufacturing and commercialization activities for MTX240 in the Licensed Territory, and Otsuka will retain all rights to MTX240 in Japan. As consideration for the license, the Company made an upfront payment to Otsuka, and Otsuka is eligible to receive one-time development and regulatory milestones, as well as tiered royalties in the mid-single digits on the net sales of licensed products. The Company is also obligated to pay Otsuka a percentage of any sublicense income it receives, subject to certain exceptions.
On 18 March 2026 the Company announced a change in the ratio of the Company’s Ordinary Shares from each ADS representing 100,000 Ordinary Shares to each ADS representing 500,000 Ordinary Shares.
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. UKNewshour.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. UKNewshour.com takes no editorial responsibility for the same.

